
Introduction to Crude Oil
Crude oil is a mixture of hydrocarbons. The petroleum as it comes from the earth may be yellow, red-brown or greenish; it is usually quite dark. It is almost entirely made up of hydrocarbons.
Carbon and hydrogen may be joined together to make many thousands of different molecules and several hundred of these are to be found in any sample of crude oil.
At school they tell you that petroleum is a mixture of alkanes but this is an oversimplification - there are three main constituents: alkanes, cycloalkanes and aromatics.
Alkanes are molecules with the formula CnH2n+2.
Cycloalkanes are cyclic alkanes; they have single bonds only and have the general formula CnH2n. They are often present in larger quanties than alkanes.
Aromatics are compounds containing benzene rings. Typical aromatic constituents of crude oil are benzene, methyl benzene and napthalene.
A fourth (minor) component is a group of compounds of indeterminate formula called asphaltics. These are large, tarry, sticky molecules, and less useful than the rest.
The composition of crude oils varies enormously depending on its source, but the % element composition is fairly constant.
COMPOSITION BY WEIGHT
83-87% Carbon
10-14% Hydrogen
up to 6% sulphur
up to 2% Nitrogen
up to 1.5% Oxygen
metals up to 0.1%
HYDROCARBONS PRESENT BY WEIGHT
........... .......... Average ...... Range
alkanes........... 30%........... 15-60%
cycloalkanes .. 49%........... 30-60%
aromatics ....... 15% ............3-30%
asphaltics ........ 6%............. the remainder
Here are some examples of compounds in crude oil:
ALKANES
pentane, hexane, octane
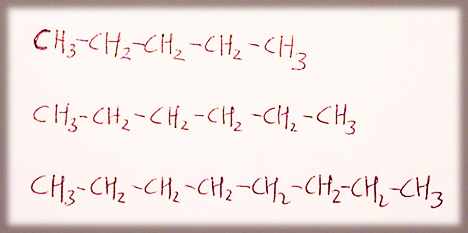
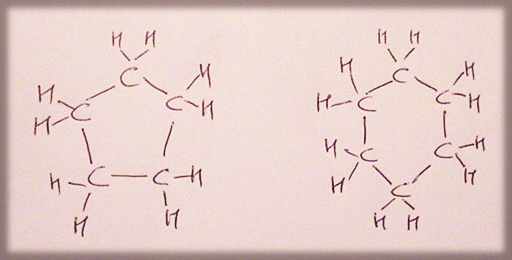
CYCLOALKANES
cyclopentane, cyclohexane
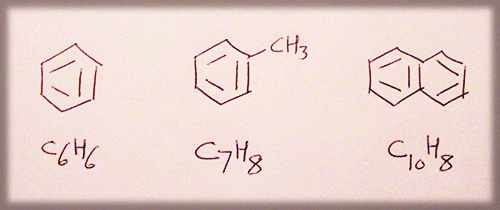
AROMATICS
benzene, toluene (methyl benzene), napthalene
Separating these compounds by fractional distillation on the basis of their boiling points creates different fuels (petrol, diesel, paraffin, etc). This is covered on a separate page.
N.D., habitat21
big turbines
small turbines